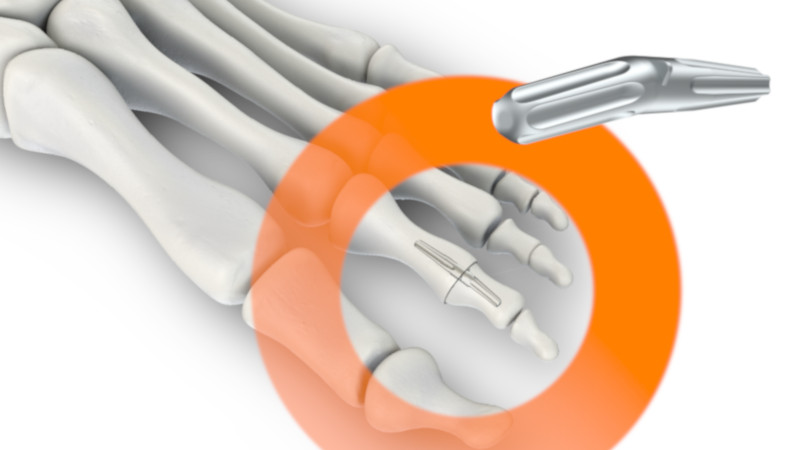
Syntellix launches StarFuse®
Syntellix AG, a leading developer and manufacturer of highly innovative, globally unique magnesium implants for orthopaedics and trauma surgery, has announced the launch of its new MAGNEZIX® StarFuse® product line. Until now, the MedTech company has mainly supplied universally applicable implants, such as compression screws and pins, and this is the first time it has launched a special implant for a strictly defined area of application.
StarFuse® implants are used to stiffen toe joints (PIP arthrodeses) in the surgical correction of so-called hammer or claw toes, which often occur in connection with a hallux valgus (bunion), from which over 10 million people in Germany suffer. With the new StarFuse® and the other proven products from its MAGNEZIX® portfolio (CS, CBS and Pin), Syntellix now offers a system solution for foot surgery to treat deformities comprehensively using advanced magnesium implants.
The new implant was developed in close co-operation with orthopaedists and foot surgeons; its highly innovative and patented design features unique stop fins and distinctive star-shaped longitudinal grooves, which enable the user to insert the implant easily and safely. It is available in three different sizes (small, medium and large), either straight (0°) or angled (10°).
Like all MAGNEZIX® implants that have been supplied so far, the StarFuse® implant also offers the special benefits of this trend-setting magnesium-based material: the material is metallic but is broken down by the body and converted into bone tissue; moreover, it has mechanical properties very similar to those of bone, such as high stability and elasticity. In addition, magnesium stimulates bone growth osteo-conductively, is infection-inhibiting and is particularly well tolerated. There are no known allergies.
Shortly after the implants became commercially available for the first time, very interested and forward-looking doctors were already using them on a regular basis in operations; in all cases, feedback was very positive. According to Dr Hubert Klauser, medical director of the HFZ Hand- und Fusszentrum Berlin, “the implants function excellently” and “are a ‘must’ for every foot surgeon”. John Göddertz, head of the Department for Foot and Ankle Surgery at the Fachklinik 360° in Ratingen, was also very satisfied with the outcome of his surgery and is already looking forward to “performing more operations”.